Ampoule And Vial Filling Machine
This integrated line is designed for the combined production processes—including washing, sterilization, filling, and sealing—of ampoules and vials in small-volume injectable workshops. (The machine can also be tested and debugged as a standalone unit.) The overall design meets advanced international and domestic standards, featuring a rational structure, high automation, stable and reliable operation, high production efficiency, and full mechatronic integration. Materials in contact with pharmaceutical liquids are made of AISI316L stainless steel, while other parts use AISI304 stainless steel. Local components are made of materials that do not contaminate the drugs or the environment. The entire design and manufacturing process complies with FDA and current GMP standards.
The integrated line consists of a vertical ultrasonic cleaning machine, a sterilization and drying oven, and an ampoule filling and sealing machine. It is equipped with coordinated linkage mechanisms and components that enable stepless speed regulation and precise control to ensure process requirements are met.
The line operates at high speed with a high yield rate, is easy to operate and maintain, and runs smoothly without impact, malfunctions, bottle jamming, or breakage.
Multiple safety devices are installed to ensure operator safety and machine integrity.
Validation ports are reserved at key locations.
The entire line is controlled by a PLC, with individual units operated via touch screens for programmed production.
Buffer mechanisms are integrated into the PLC program at the connection points between machines, ensuring reliable and stable operation.
The line is equipped with a "no bottle, no filling" function.
A centralized lubrication system is included to simplify oiling at various lubrication points.
The safety system of the entire line meets international safety standards. Operation and mold replacement are straightforward, and maintenance is convenient.
The machine features alarm and fault display functions.
Ampoule-Vial Dual-Purpose Integrated Production Line: Maximum Flexibility for Sterile Manufacturing
Introducing a new paradigm in pharmaceutical production: the Ampoule-Vial Dual-Purpose Integrated Production Line. This innovative system eliminates the need for separate, dedicated equipment by seamlessly handling both glass ampoules and a wide range of vials (including serum and lyophilization vials) on a single, unified platform. Engineered for facilities that require agile, multi-format aseptic production, this line delivers unparalleled flexibility without compromising the sterility or precision required for injectable drugs.
At the core of this revolutionary line is a highly adaptable ampoule and vial filling machine. This sophisticated piece of engineering can be quickly reconfigured to switch between the hermetic sealing of ampoules and the stoppering and capping of vials. Whether you need a high-speed vial filling machine for batch processing or a versatile system for diverse product portfolios, this line is the ultimate solution.
Key Features & Benefits:
-
Dual-Format Agility: Dramatically reduce changeover times and capital expenditure. This single line empowers you to produce both ampoules and vials, allowing you to respond swiftly to market demands and optimize your production schedule for maximum asset utilization.
-
Uncompromised Aseptic Integrity: Designed as a fully integrated pharmaceutical vial filling machine, the line maintains a continuous, validated sterile environment from container washing through to final sealing. It incorporates advanced sterilization tunnels and a Class A protective environment at the critical filling and sealing zone.
-
Precision Filling & Sealing: The heart of the system is the versatile ampoule and vial filling machine. It ensures pinpoint volumetric accuracy for even the most sensitive formulations. The machine intelligently adapts its process—performing a pull-seal for ampoules and applying a stopper and overseal for vials—all with flawless execution.
-
Robust Pharmaceutical-Grade Construction: Built to the most stringent cGMP standards, this pharmaceutical vial filling machine is constructed from 316L stainless steel and other compliant materials. It is designed for easy cleaning, sterilization, and long-term reliability in a demanding production environment.
-
Future-Proof Your Investment: This line is more than just a vial filling machine; it is a strategic asset. Its inherent flexibility protects your investment against changing product pipelines and packaging trends, ensuring your manufacturing capability remains state-of-the-art for years to come.
Ideal for:
-
Pharmaceutical Contract Manufacturing Organizations (CMOs)
-
Biotech companies with diverse injectable product portfolios
-
Production of vaccines, antibiotics, hormones, and lyophilized powders
-
Any facility requiring high-mix, low-volume or high-volume sterile production
Consolidate Your Capabilities. Multiply Your Potential.
Stop managing multiple, single-format lines. Consolidate your ampoule and vial production into one efficient, reliable, and future-proof system. This integrated line, featuring a world-class ampoule and vial filling machine, provides the flexibility of a specialized vial filling machine with the added capability for ampoule production.
Upgrade to the ultimate pharmaceutical vial filling machine solution. Contact us today to discover how our dual-purpose technology can transform your sterile manufacturing operations.
| Ampoule process flow |
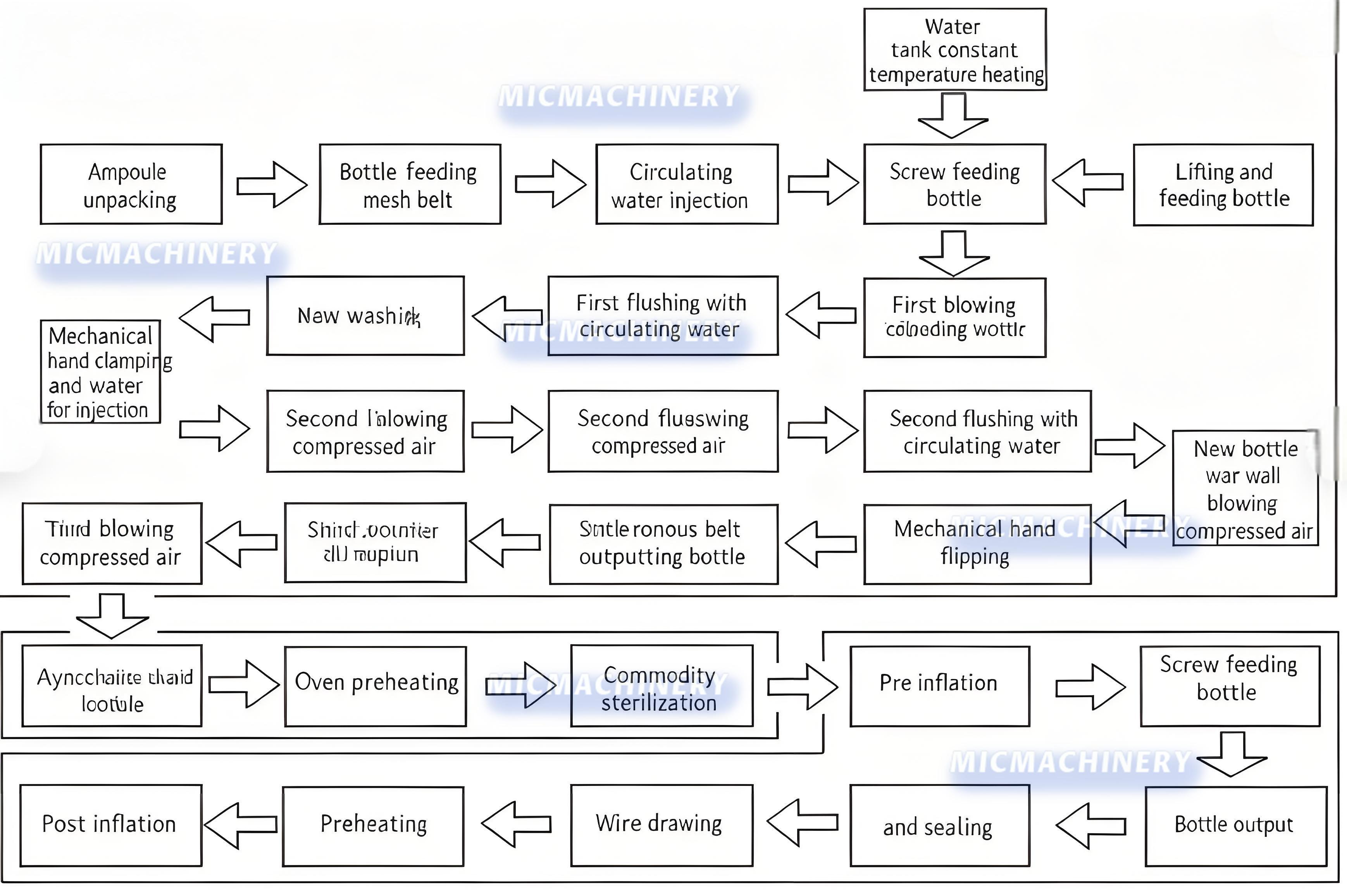 |
|
vial process flow |
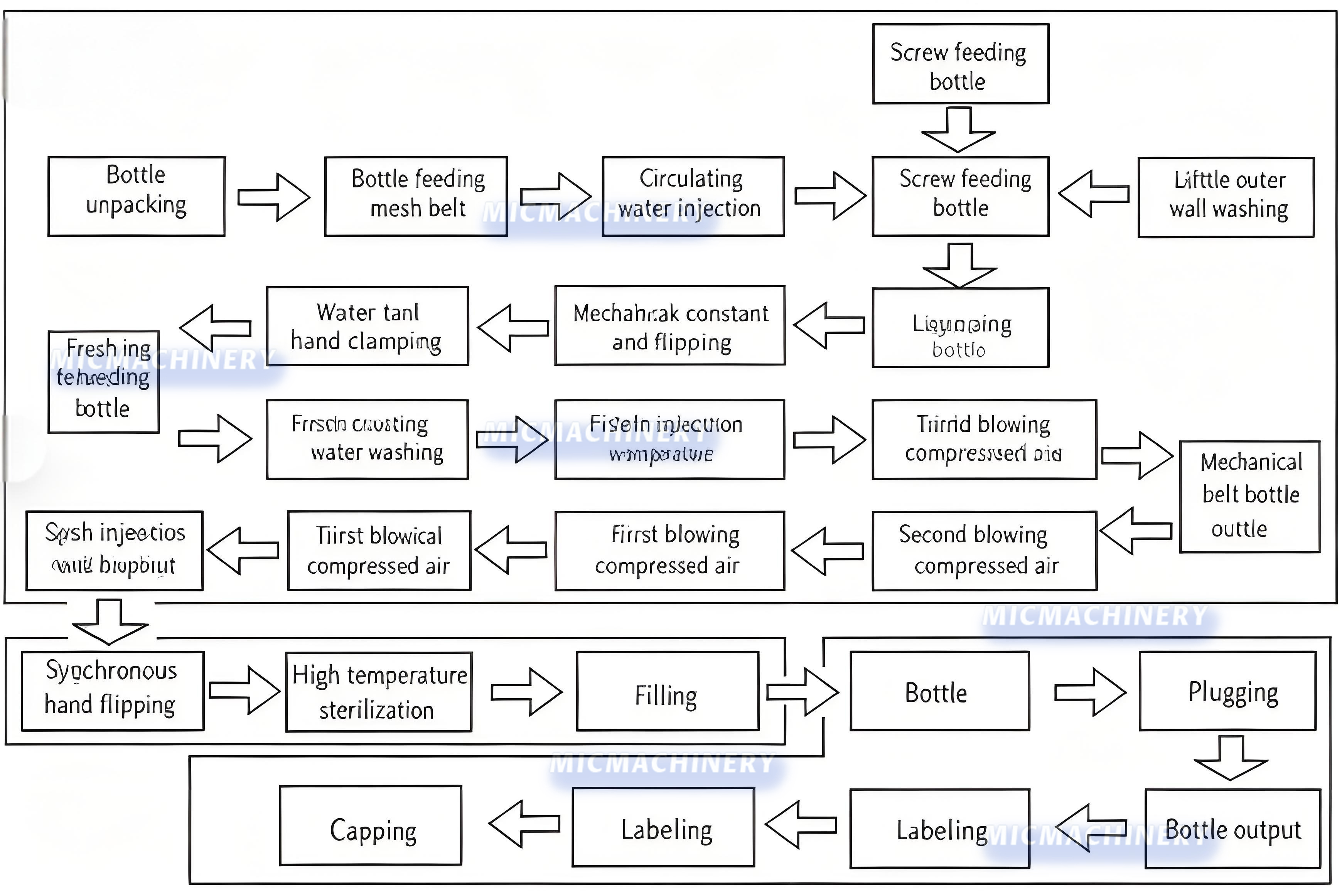 |
Technical parameters of bottle washing machine
| Parameter | Specification |
|---|---|
| Production Capacity | Ampoule: 400 bottles/min Vial: 250 bottles/min |
| Suitable Specification | Ampoule: 1ml-10ml Vial: 8-15ml |
| Breakage Rate | ≤ 0.1% |
| Cleaning Quality (Clarity) | No visible white particles, color spots, fibers, or glass fragments; other visible foreign matters < 1 per bottle. |
| Gripping Error Rate | ≤ 0.05% |
| Bottle Output Counting Error | ≤ 0.01% |
| Residual Water | 0.1g (measured by drying method) |
| Visual Inspection Pass Rate | 100% |
| Water Consumption | 0.4-0.6 m³/h (at pressure ≤ 0.2-0.3 MPa) |
| Compressed Air Consumption | 40-50 m³/h (at pressure ≤ 0.25-0.35 MPa) |
| Power Capacity | 15 KW, 380V |
| Dimensions | 2000 × 2204 × 1350 mm |
| Weight | 2400 KG |
Technical parameters of hot air circulation oven
| Parameter | Specification |
|---|---|
| Suitable Specification | 1-20ml Ampoules/Vials |
| Production Capacity (Ampoule) | 400 bottles/min (1-20ml) |
| Effective Conveyor Belt Width | 620 mm |
| Sterilization Temperature | 280°C - 320°C |
| FH Value | > 1365 |
| Sterilization Time | ≥ 5 min |
| Temperature Probe Accuracy | ≤ ±2°C |
| Temperature Distribution (Empty, Sterilization Section) | ≤ ±5°C |
| Temperature Distribution (Loaded, Sterilization Section) | ≤ ±7.5°C |
| Bottle Outlet Temperature | ≤ Ambient Temperature + 15°C |
| Exhaust Air Volume | 3000 - 4100 m³/h |
| Power Capacity | 71 kW (including 60 kW for electric heating) |
| Overall Dimensions | 4538 × 1465 × 2445 mm |
| Net Weight | 3500 kg |
Main specifications and technical parameters of the capping machine
| Parameter | Specification |
|---|---|
| Bottle Specification | 8-15ml Vial (Requires parts changeover) |
| Production Capacity | 250 bottles/min (Adjustable) |
| Number of Capping Heads | 10 heads, 3 rollers each |
| Capping Qualification Rate | ≥ 98% |
| Bottle Breakage Rate | ≤ 0.01% |
| Oscillator Power | 40 - 120 W |
| Total Power | 2.5 kW |
| Machine Weight | 1500 Kg |
| Overall Dimensions | 1300 × 2300 × 2450 mm |
Please feel free to let us know if you need any help, we will provide a professional guidance for you! Contact Us

