Overview of the Aseptic Blow-Fill-Seal System

The Aseptic Blow-Fill-Seal (BFS) System is an advanced pharmaceutical manufacturing technology that integrates container forming, sterile filling, and sealing into a single automated process. By eliminating unnecessary human contact and minimizing contamination risk, BFS systems ensure the safe production of sterile ophthalmic solutions, inhalation products, infusion containers, and small-volume liquid preparations in compliance with global GMP requirements.
⚙ How Does the Blow-Fill-Seal Process Work?
| The BFS process begins with the extrusion of pharmaceutical-grade resin, which is molded into a sterile container. Without leaving the isolated chamber, the newly formed container is filled and sealed within seconds. Intelligent PLC controls, aseptic airflow, precision temperature balancing, and automated inspections work together to maintain stable, repeatable, and contamination-free operation suitable for sensitive medical and pharmaceutical applications. |
|
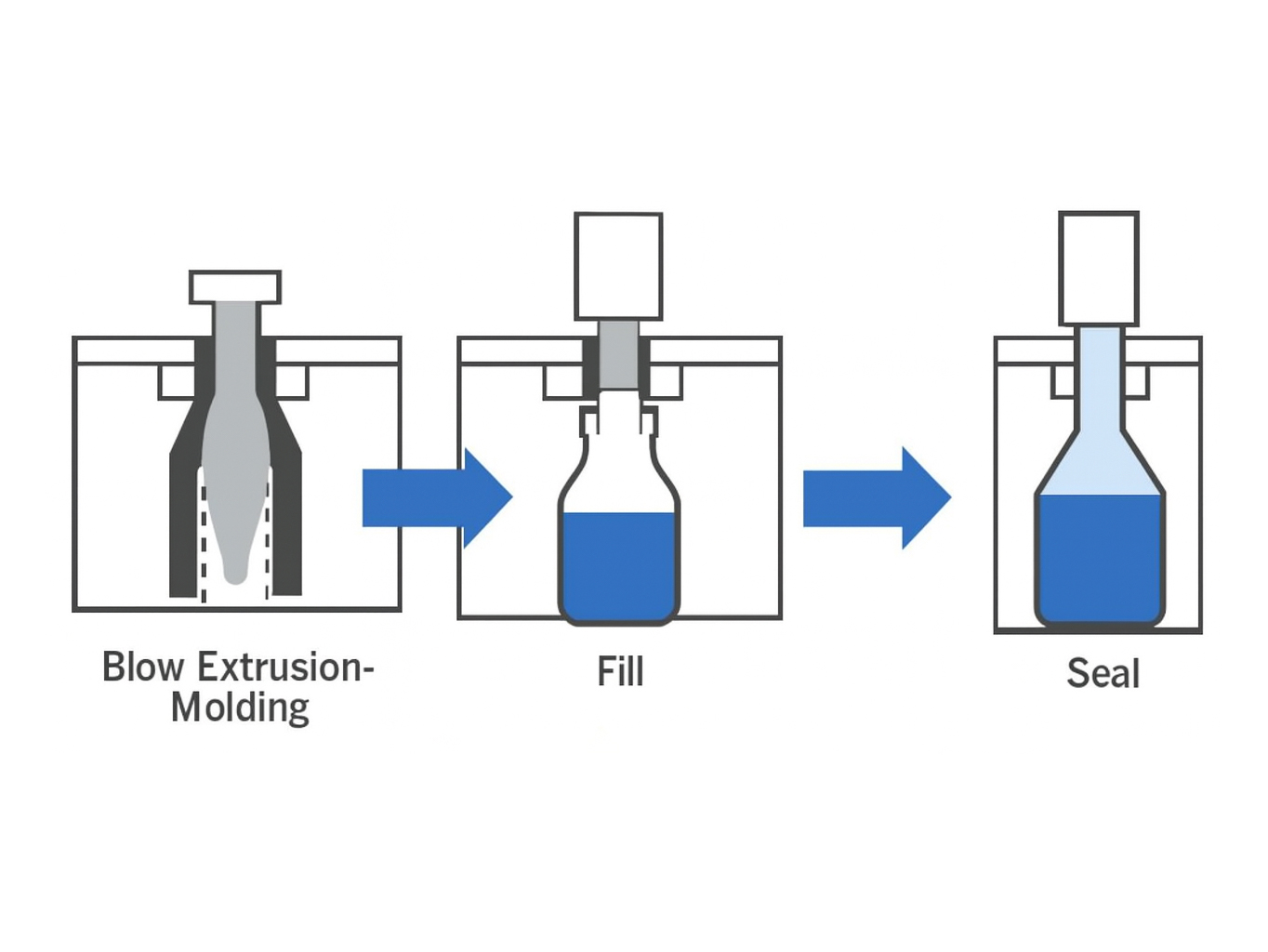 |
⚙ Core Components and System Features
 |
|
Aseptic Blow-Fill-Seal Systems from Mic Machinery are engineered with a series of high-performance modules designed to support reliable, efficient, and contamination-free production on a continuous basis:
-----
· Container Molding Unit: The resin extrusion and molding system forms containers such as ampoules, vials, and bottles directly within the sterile zone. This eliminates the need for pre-made containers and ensures absolute purity, structural consistency, and high-speed molding capability for various pharmaceutical formats.
· Aseptic Filling Module: Designed for maximum dosing accuracy, this module supports a wide range of formulations, including ophthalmic solutions, inhalation drugs, infusion liquids, and biotech products. Advanced dosing pumps, flow meters, and sterile nozzles ensure each container receives the correct volume under highly controlled conditions.
· Sealing and Quality-Control Systems: High-temperature sealing ensures container closure integrity, while in-line inspection technology performs leak detection, visual checks, and dimensional verification. These systems work seamlessly to maintain sterility, guarantee product quality, and reduce the risk of defective batches in demanding pharmaceutical environments.
|
⚙ Where Can BFS Technology Be Applied?
BFS technology is used in pharmaceutical factories, hospital preparation units, biotech laboratories, and diagnostic solution facilities. It is particularly suited for producing sterile ophthalmic products, respiratory medications, pediatric solutions, infusion additives, and single-dose preparations. Because the entire process takes place in a protected environment, BFS supports large-scale production for commercial use as well as small-batch formulations for clinical testing or specialty medical applications.
⚙ Primary Application Scenarios and Advantages
|
Mic Machinery’s BFS Systems offer broad compatibility with various pharmaceutical and clinical production needs. Their reliability and precision make them suitable for:
-----
· Ophthalmic and Respiratory Products: Highly sterile eye drops, nasal preparations, and inhalation medications, where microbial control, particle-free packaging, and container integrity are crucial for patient safety and product stability. The BFS method ensures each container is produced in a controlled environment with minimal exposure to external air, guaranteeing superior sterility.
· Infusion and Clinical Liquids: Ideal for producing non-PVC infusion bags, pediatric formulas, and emergency-dose sterile liquids. The ability to form, fill, and seal containers in a single chamber accelerates production while ensuring uniform dosage accuracy and regulatory compliance for hospital supply chains.
· Biopharmaceutical and Laboratory Use: Widely used for experimental solutions, research-grade liquids, diagnostic samples, and single-dose units required for scientific studies. BFS ensures consistent sterility, reproducibility, and packaging durability, making it suitable for sensitive formulations and precise laboratory workflows.
|
|
 |
⚙ What After-Sales Services Does Mic Machinery Provide?
Mic Machinery ensures long-term system performance with comprehensive after-sales support designed for high-demand pharmaceutical operations:
· Installation, Commissioning & Qualification: Expert technicians handle on-site installation, complete machine setup, tune the production parameters, and perform IQ/OQ/PQ validation to meet strict GMP and regulatory standards.
· Training & Maintenance: Mic Machinery provides full operator training, maintenance planning, scheduled inspections, and continuous technical guidance. These services help clients maintain stable production, reduce downtime, and improve operational efficiency.
· Technical Assistance & Upgrades: A 24/7 remote support system, rapid troubleshooting, spare-part availability, and customizable upgrades ensure equipment longevity. Clients can receive tailored enhancement packages to support capacity expansion or new product development.
⚙ Mic Machinery – Your Global Partner in BFS Technology
Mic Machinery is a trusted manufacturer specializing in pharmaceutical sterilization and packaging equipment, including advanced Aseptic Blow-Fill-Seal Systems. With strong capabilities in engineering design, precision manufacturing, and quality assurance, the company delivers solutions that meet global GMP requirements and evolving industry standards. From initial consultation and project planning to installation, user training, and long-term service support, Mic Machinery assists clients worldwide in building highly efficient sterile production lines. The company’s commitment to innovation, reliability, and customer satisfaction ensures that every BFS-related project achieves safe, compliant, and sustainable manufacturing performance.