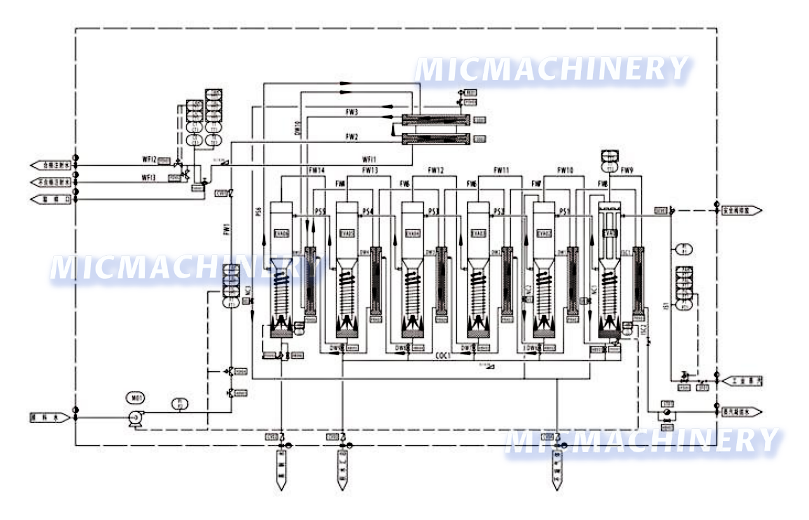
Water for injection generation and storage system
The water for injection generation and storage system uses SS316L material, adopts multi-effect distillation + three-stage separation, and features double tube sheets and argon-protected welding. With HMI+PLC full automation, online parameter monitoring, and audit trail function, it meets GAMP 5 standards, ensuring high-purity, contaminant-free injection water supply.
feature
- Water for injection generation and storage system ensures safe, compliant water production and supply.
- Its structure features double-tube-sheet design for the external preheater, condenser, and first-effect evaporator (inner sheet expanded and welded externally to prevent cross-contamination).
- A three-stage separation device (falling film evaporation + gravity + spiral flow) refines the process.
- All raw water, product water, and secondary-steam contact parts use SS316L.
- The evaporator, preheater, and condenser rely on aluminum silicate needle-punched blankets for thermal insulation (good extensibility, light weight, etc.).
- Electrochemical polishing smooths internal and external surfaces.
- Energy-saving multi-effect distillation reuses secondary steam step-by-step, cutting industrial steam use and cooling water (five-effect needs little, six-effect none).
- Piping uses stainless-steel elbows (minimizing welding); welded parts get argon-protected automatic welding (single-side welding, double-side forming) plus post-weld electrochemical polishing.
- Verification materials (endoscopy photos, etc.) prove quality.
- The HMI+PLC control system enables full automation, simple operation, multi-communication modes, and at least three authority levels.
- Online monitoring tracks conductivity, temperature, and flow (paperless/paper recorders optional).
- Optional electronic signatures/records create audit trails, meeting GAMP 5 and 21 CFR PART 11.
- Ideal for wholesale water for injection generation and storage system business, it guarantees reliable water for injection generation and storage system supply.
System advantage
- Strong Compliance: It adopts SS316L contact material, double tube plate structure, and argon arc welding process. The produced water meets the standards of Chinese Pharmacopoeia (CP2020), United States Pharmacopoeia (USP43), and European Pharmacopoeia (EP10), satisfying GAMP 5 and 21 CFR PART 11 compliance requirements, thus adapting to the strict regulatory needs of the pharmaceutical industry.
- High Water Purity: Through three-stage separation technology of falling film evaporation + gravity + cyclone, combined with electrochemical polishing surface treatment, it avoids cross-contamination and microbial growth, ensuring the high purity of injection water.
- Energy-Saving and Efficient: Multi-effect distillation recovers secondary steam step by step. The more evaporators there are, the more significant the energy-saving effect (six-effect distillation does not require cooling water), greatly reducing industrial steam and cooling water consumption, and lowering operating costs.
- High Automation and Safety: HMI + PLC full automation control supports three-level permission management, online monitoring of conductivity / temperature / flow, and electronic signature audit trail, making operation convenient and data traceable.
- Stable and Durable: The aluminum silicate needle-punched blanket insulation layer has both heat insulation and stability. The pipeline adopts a design with fewer welds + argon arc protected welding, extending the equipment service life and adapting to long-term continuous operation scenarios.
Applications
- Pharmaceutical Industry: Used for mixing, dissolving, cleaning, and sterilization in the production processes of injectables, biologics, vaccines, etc. It is a core equipment for GMP-compliant production in pharmaceutical enterprises and a major application scenario for the generation and storage system of water for injection.
- Healthcare Industry: Suitable for medical instrument sterilization water in hospital operating rooms and disinfection supply centers, as well as scenarios such as clinical intravenous infusion and drug dilution, ensuring medical water safety and supporting the healthcare-end demand for the water for injection generation and storage system supply.
- Biotechnology Field: Meets the strict requirements for sterile high-purity water in bioreactor cultivation and cell product manufacturing, providing stable water quality assurance for biotechnology enterprises.
- High-End Laboratories: Serves precise experiments such as drug R&D and clinical testing, providing standard-compliant laboratory water to ensure the accuracy of experimental results.
- Chemical and Electronic Industries: Used in scenarios such as high-purity chemical raw material synthesis and electronic component cleaning, adapting to industrial production links with high requirements for water purity.
| Capacity(L/h) | Industrial steam consumption(L/h) | Feed water consumption(L/h) | Cooling water consumption(L/h) | Dimensions(mm) | |
| LDS100 - 4 | ≥100 | 30 | 110 | 80 | 1080×585×2800 |
| LDS300 - 5 | ≥300 | 80 | 330 | 170 | 1760×755×3000 |
| LDS500 - 5 | ≥500 | 115 | 550 | 190 | 2000×805×3500 |
| LDS1000 - 5 | ≥1000 | 250 | 1100 | 350 | 2300×1000×4000 |
| LDS1000 - 6 | ≥1000 | 210 | 1100 | 0 | 2930×1000×4000 |
| LDS2000 - 6 | ≥2000 | 440 | 2200 | 0 | 3650×1200×4100 |
| LDS3000 - 6 | ≥3000 | 670 | 3300 | 0 | 3800×1200×4200 |
| LDS4000 - 6 | ≥4000 | 900 | 4400 | 0 | 4300×1250×4500 |
| LDS5000 - 6 | ≥5000 | 1130 | 5500 | 0 | 4600×1400×4600 |
| LDS6000 - 6 | ≥6000 | 1350 | 6600 | 0 | 4600×1400×4900 |
contact us
Please feel free to let us know if you need any help, we will provide a professional guidance for you! Contact Us




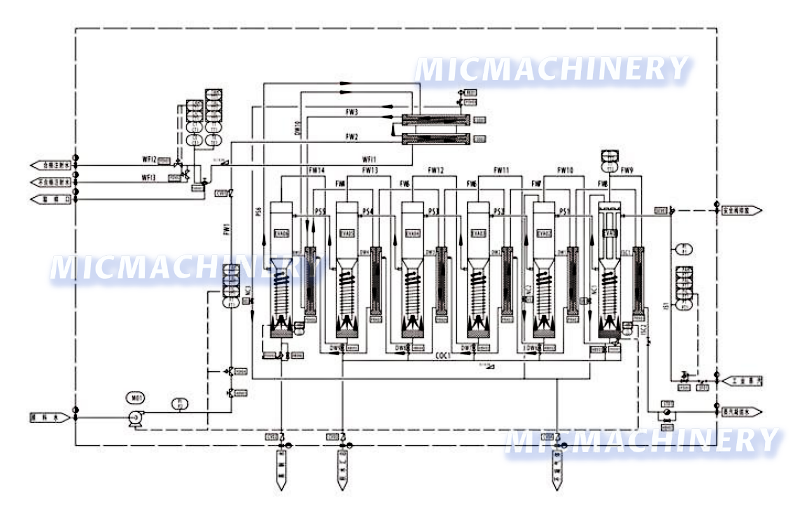
 Water for injection generation and storage system
Water for injection generation and storage system